INTRODUCING
RecoveryRx
MEETING THE NEED FOR
an effective, drug-free, analgesic therapy to limit the negative consequences of under-managed pain.
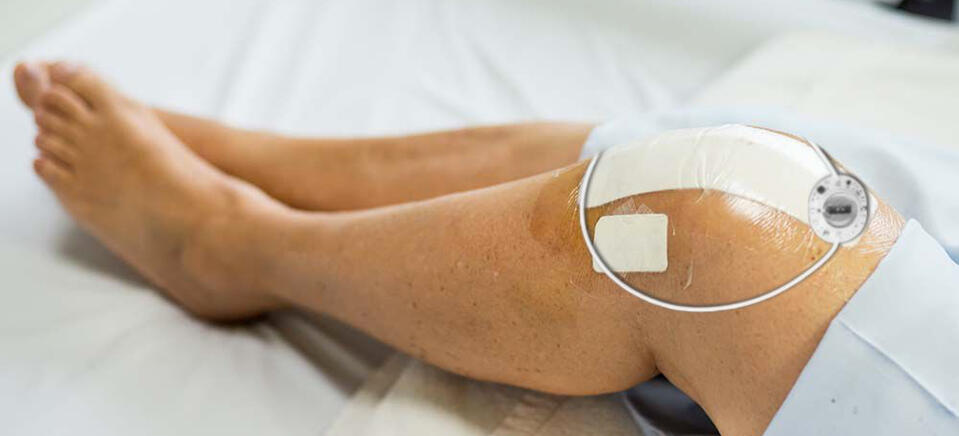
What is RecoveryRx?
An innovation in drug-free pain and edema management. RecoveryRx is a class II, USA FDA-cleared, non-thermal shortwave therapy device which utilizes radio-frequency electromagnetic energy to modulate nerve activity. When used adjunctively, RecoveryRx has been found to reduce pain and the consumption of pain medication, including opioids. RecoveryRx is non-invasive and has no known side effects. Each unit delivers 720 hours of paresthesia-free therapy, allowing for 24/7 use.

FDA cleared
Drug-free-no risk of adverse interactions
Paresthesia-free, can be used during sleep
Lightweight, wearable device- can be used while ambulatory
Affordable
NO known side effects
Reduces and often eliminates the need for pain medications- including opioids
Easily applied by you or the patient for at home therapy
Listed on DAPA Part# 277-4
Most Recent Case Synopses
"These cases demonstrate that following knee and hip arthroplasty, the use of pulsed shortwave therapy devices is feasible in both the in- and out-patient settings and may improve analgesia while decreasing—perhaps sometimes obviating—opioid consumption." Full article here:https://www.fortunejournals.com/articles/noninvasive-wearable-pulsed-shortwave-radiofrequency-therapy-for-analgesia-following-knee-and-hip-arthroplasty-a-case-series.pdf"These cases suggest that pulsed electromagnetic field therapy may be an effective treatment for intractable postamputation pain. Considering the low patient burden of noninvasive, wearable devices, combined with few contraindications and no significant side effects or adverse events, further study with a randomized, controlled trial is warranted." Full article here:https://amjcaserep.com/abstract/full/idArt/937549"Following moderately painful ambulatory umbilical (n=3) and inguinal (n=2) hernia repair as well as bilateral breast surgery (n=2),patients had taped over their incision(s) 1 or 2 nonivasive, wearable, disposable, pulsed shortwave therapy devices (RecoveryRx)which functioned continuously for 30 days. Average resting pain scores measured on the 0-10 numeric rating scale were a median of 0 during the entire treatment period. Six patients avoided opioid use entirely, while the remaining individual required only 5 mg of oxycodone during the first postoperative day."
Article here: https://pubmed.ncbi.nlm.nih.gov/36463434Brian M Ilfeld, Brannon J Cha, Andrew S Qian, John J Finneran, Engy T Said and Baharin Abdullah-Treating intractable postamputation pain with wearable, non-invasive, non-thermal, pulsed shortwave (radiofrequency) therapy: a randomized, double-masked, sham-controlled, crossover pilot study. Read the full article here: https://rapm.bmj.com/content/early/2024/02/21/rapm-2023-105154?fbclid=IwAR2m5KaSiMecct7QFGHrRyDtdU4c6DF0CesQPel4C50_TNxHlD0vpoIg4

Frequently Asked Questions
How does the device work?Pain occurs when the nervous system sends signals to the brain arising from the injury caused by surgery. The device alters the way the brain interprets these pain signals, thereby increasing the individual’s pain threshold, i.e., reducing the person’s perceived pain The device can be used 24/7.Is it safe to use?The device is drug free and has no harmful side effects. The device can be worn by patients with fragile skin.
Note: The device is nonsterile and should not be applied directly over open wounds, however it can be applied over bandaged wounds.Is the device clinically proven?Yes. The device has been clinically proven through a series of clinical studies, of which more are ongoing, at leading medical institutions. The technology has been used for decades in hospitals and clinics and has received an overwhelming amount of positive testimonials from consumers.What will your patient feel?ONLY BETTER! The patient will not feel heat nor will they feel the low level energy that is gently pulsed into the nerves.How long until the patient feels pain relief?Depending on the severity of the injury, patient pain levels can begin to subside after only 2-3 hours of wearing the device and will continue to decrease as long as the device is being used continuously or at least 12 hours per day. However, in some instances it could take up to 3-4 days for the therapy to take effect.Is the RecoveryRx device a magnet?No. It is a battery operated pulsed electromagnetic medical device. When activated, the devices gently pulse a low level energy into the injured area reducing pain and inflammation.How long do these products last?The RecoveryRx® products last for 720 hours and can be turned on and off as needed.How does the device stay on the body?The RecoveryRx® product includes flexible and comfortable light adhesives to be used over the wound dressing. The patient could also use medical grade tape to adhere to your body.Where should the patient place the device for maximum treatment?The therapeutic area is within the loop of the device. The loop area should be placed directly over the source of the pain.Can the patient wear the device on other areas of the body?Yes. These devices are also effective in treating musculoskeletal pain. The patient can use any medical grade adhesive or wrap to adhere the device to the area in pain.How close to the body should the patient wear the device?The device should be placed as close to the skin as possible. The therapy will also be effective through light clothing. If the medical adhesive strips are too harsh on the skin, they may apply the device over spandex or tight fitted clothing. Use medical tape or wrap to adhere the device to the shirt over the source of the pain.Can the patient wear more than one device?Yes. You they can wear more than one medical device at a time as long as none of the devices are on top of another.Can the RecoveryRx® product be placed under compression dressings?No. RecoveryRx® can’t be used under compression dressings; however, the device can be used over the compression dressing with the energy passing through to the tissue below.Does it matter what direction the device is placed on the body?No. The module only houses the battery and can be placed in any direction. What is important is the device loop should be placed directly over the source of the pain for maximum treatment.Can I replace the battery?No. These devices are disposable and should be discarded when the battery life has ended. Engineers determined that the device is no longer able to give an optimal treatment after 720 hours because of continued bodily movement which causes wear on the internal components. For this reason, the device needs to be discarded after the operation time. These can be disposed of in ordinary trash. There are no user-serviceable parts inside the unit. Do not attempt to modify or break open the device.Are these safe to use with metal implants and pacemakers?The RF field strength is too low for any significant heating from implanted metal so it is safe for all metal implants. There is no concern for pacemakers as they have built in RF protection. Though as a precaution, do not use this device directly over a cardiac pacemaker, implanted defibrillator, deep brain stimulator and nerve stimulators or other active implantable device.Is the RecoveryRx device waterproof?This device is not waterproof and therefore should not be submerged under water.Can pregnant women use the device?Our products have not been clinically evaluated for use during pregnancy; therefore, we cannot recommend its use.
© Untitled. All rights reserved.